MGC Pharmaceuticals has expanded the number of patients prescribing for or being processed to receive prescriptions of its medical cannabis-based pharmaceutical products in Australia and the UK to around 500 with the company racking up its 1000th prescription this month. Management said that the key milestone readily demonstrates its strengthening position and momentum in those key markets, with its sales graph continuing to climb month by month.

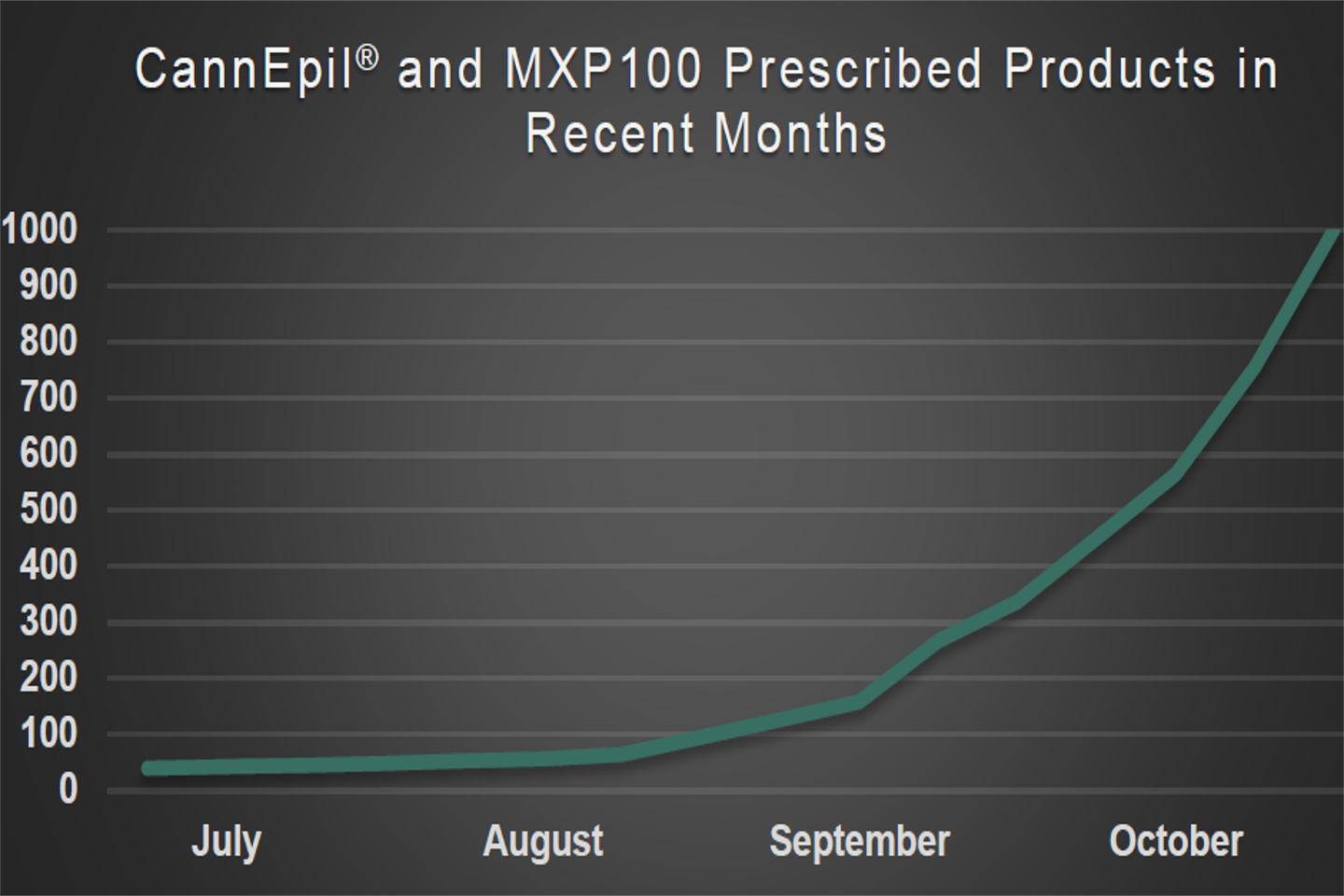
MGC Pharmaceuticals has achieved a significant milestone, this week surpassing more than 1,000 prescribed products to more than 500 unique patients that have either prescribed for or are being processed to receive prescriptions of its medicinal cannabis-based pharmaceutical products in Australia and the UK.
Notably, the company more than doubled the number of units prescribed in October, when compared to September.
Management said the recent landmark milestone appears to underline its strengthening position and the rapidly growing popularity of medicinal cannabis-based health products.
MGC has managed to grow the demand for its proprietary medicines on a global scale within a relatively short period, demonstrating the immediate, ongoing and material revenue-generating potential of the company’s products.
These include CannEpil and MXP100, which are prescribed in Australia under the Special Access Scheme and in the UK through specialised doctors permitted to prescribe phytocannabinoid-derived medicines under the Early Access to Medicines Scheme.
CannEpil is the company’s high cannabidiol, or “CBD”, and low tetrahydrocannabinol, or “THC”, phytocannabinoid-derived formulation, used in the treatment of drug-resistant epilepsy.
The MXP100 product is recommended for use in mild neurological disorders, inflammatory and hepatoma patients, with the latter being a cancer of the cells of the liver.
Both products are proprietary medicinal drugs developed internally through MGC’s research and development divisions.
The company is rapidly advancing its international distribution efforts in a bid to lock down major markets ahead of any competing products.
One of MGC’s key distribution partners, Cannvalate, who account for more than 50% of prescriptions to date, due to its close ties with the medical community and national footprint, has reported increased interest in the company’s products.
MGC also said that Cannabis Access Clinics had recently joined fellow Australian, Tetra Health, as one of the company’s important clinical partners who will expand the network of medical professionals informed of its available product range.
The company’s European-based, “seed to pharmacy” strategy closes the loop for its medicinal cannabis products, with the MGC now on track to significantly increase its production capacity after recently signing a long-term lease agreement with Maltese Government-owned Malta Industrial Parks, for a 6,000 square metre site to cultivate cannabis plants.
The company has begun site works for the fully-integrated, multi-storey building in Malta that will contain 15,000 square metres of space for the cultivation and manufacture of cannabinoid-derived medicines for its proposed global distribution networks.
This facility, when constructed, will be capable of producing over 8,000 units of each MGC Pharma product per hour and has been designed with significant excess capacity to meet the company’s future needs, once in full swing.
MGC’s “EU-GMP” manufacturing facility in Slovenia can produce pharmaceutical-grade phytocannabinoid medicines for about 5,000 patients a month at full capacity.
The ASX-listed company recently received new purchase orders exceeding €0.3m, or AUD$485,000, across its product range, as demand continues to skyrocket globally.
MGC Pharma Co-founder and Managing Director, Roby Zomer said: “From a standing start, we have now hit 1,000 prescriptions in a short space of time and ahead of schedule, demonstrating a clear demand for MGC Pharma’s products. The established profile of return patients for (the) products is expected to drive the prescription growth rate through November, December and beyond.”
Cannvalate CEO Sud Agarwal said: “We are delighted to see that MGC Pharma's product range has been so well received by the medical community. Each week, there are hundreds of patients that are accessing MGC range medication via our network. The spike of interest has been due to superior quality (EU-GMP), competitive pricing and individually designed condition-specific medications.”
NB: This article is for general financial markets news purposes only and is not to be taken as an endorsement of, or advertisement for any individual product, medicine or drug.
Is your ASX listed company doing something interesting ? Contact : matt.birney@businessnews.com.au













