Positive new data has renewed the confidence of cell therapy pioneer Chimeric Therapeutics in its pathway towards treating multiple types of cancer with its next-generation armoured natural killer (NK) cell platform, “CHM0301”.
The platform, which is being evaluated in its first clinical trial, has shown positive in vitro results in modelling human acute myeloid leukaemia (AML) and colorectal cancer (CRC) by overcoming the resistance created by a tumour’s microenvironment.
Chimeric says a key factor is a protein known as “TGFβ” – or transforming growth factor beta – which it describes as a key immune-suppressive factor known for its ability to inhibit anti-tumour medication. TGFβ has been linked to tumour invasion, metastasis, immune evasion and poor prognosis across multiple cancer types.
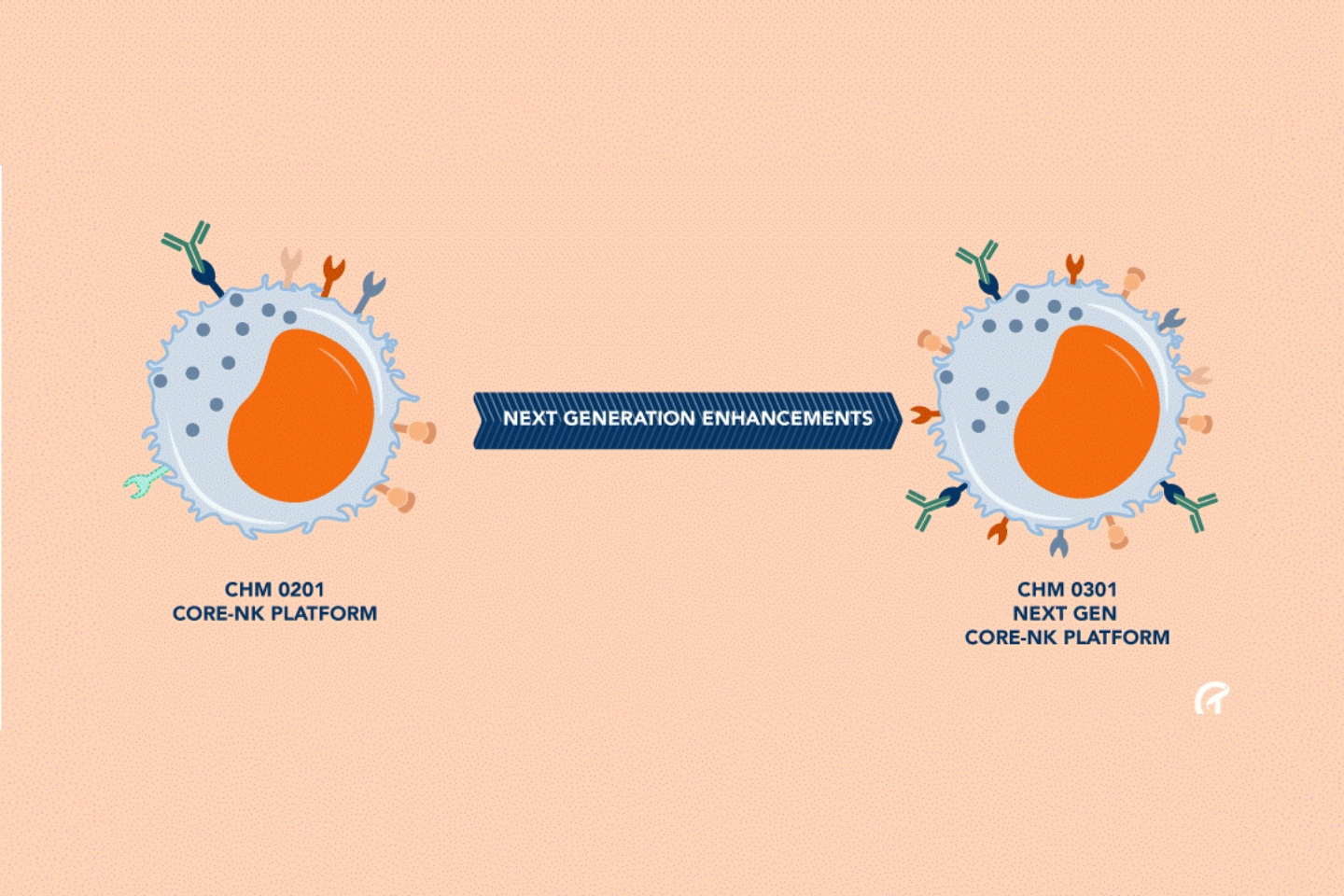
But the company says its CHM 0301 has “armouring” enhancements to enable cells to overcome immune-suppressive tumour micro-environments expressed by the TGBF factor. In vitro models of human AML and CRC demonstrated significant enhancement of TGBF resistance and potency compared to first generation CHM 0201 cells – the platform on which CHM 0301 was designed.
Chimeric initiated a sponsored research collaboration with a laboratory led by Dr David Wald, who invented the CHM 0201 NK cell platform, in November last year. Within six months, Dr Wald and his team had designed, produced and characterised CHM 0301 to maximise its potency and increase the resistance of CHM 0201 NK cells to TGFβ.
“The activity of CHM 0301 is highly encouraging and provides compelling support for this platform as the foundation for a new generation of NK cell therapies,” Dr Wald said.
Chimeric Therapeutics chief business officer and head of external Innovation Eliot Bourk said the company was encouraged by the progress of the collaboration with Dr Wald and the “highly-promising” in vitro results to date.
“We look forward to further advancing our next-generation armoured NK platform and initiating work on our lead CAR (Chimeric Antigen Receptor) NK programs for solid tumours as we continue our collaboration,” he said.
It appears Chimeric is continuing to enhance its promise of life for more patients with cancer through its innovative discovery and development of new cell therapies.
Is your ASX-listed company doing something interesting? Contact: matt.birney@businessnews.com.au