A protein derived from scorpion toxin being used to spearhead Chimeric Therapeutics’ assault on brain cancer cells has delivered encouraging early results from a study being conducted at the world-famous City of Hope Hospital in Los Angeles. Patients in the study responded well to the simultaneous introduction of the cell therapy by injecting it into both the tumours and the brain.

A protein derived from scorpion toxin being used to spearhead Chimeric Therapeutics’ cell therapy assault on brain cancer cells has delivered encouraging early results from a study being conducted at the world-famous City of Hope Hospital in Los Angeles.
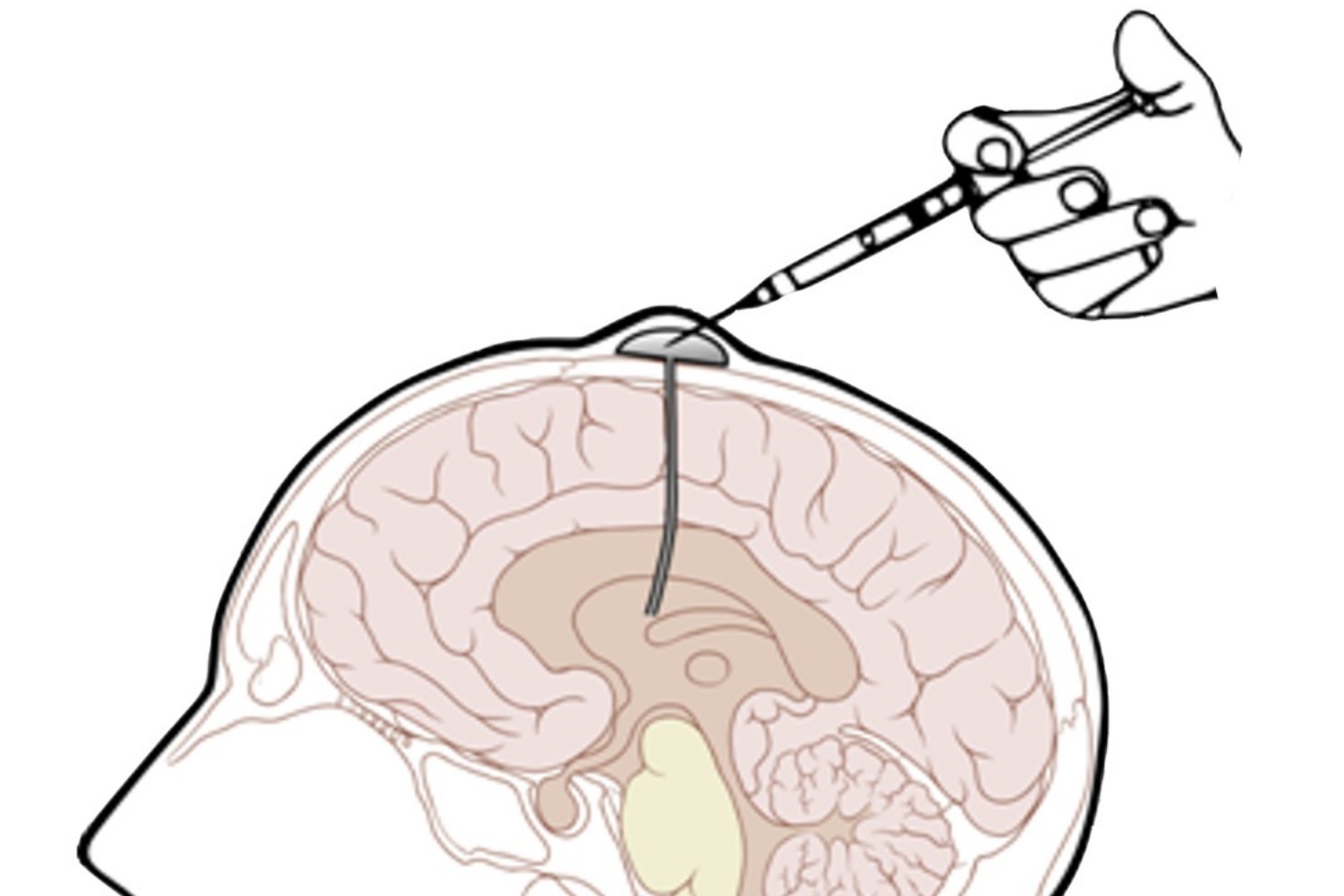
Patients in the study responded well to the simultaneous introduction of the ASX-listed company’s cell therapy that was targeted directly at their tumours and brain as part of Phase one clinical trials.
This is the second round of trials and uses a stronger dosage of chlorotoxin, a protein derived from scorpion toxin, or CLTX. The dose escalation study on patients with recurrent/progressive glioblastoma was carried out at the City of Hope, one of the largest cancer research and treatment organisations in the United States.
Glioblastoma is an aggressive type of cancer that can occur in the brain or spinal cord and while relatively rare, is almost always lethal. With less than five per cent of patients surviving for more than five years.
The company said positive feedback on side effects was reported with patients generally tolerating the dual pathways of the therapy’s administration.
All patients advanced past the 28-day follow-up without experiencing dose limiting toxicities, with two of the three patients treated in this second cohort achieving a local stability of disease.
Chimeric and City of Hope are now enrolling patients in the third cohort that will administer the protein enhanced T cells to patients through dual target delivery at an increased dose of more than double that of the second round.
The company’s therapy uniquely utilises CLTX as the tumour-targeting component of the chimeric antigen receptor. In chimeric antigen receptor treatment, a patient’s own T cells are taken from their body and ‘trained’ before being put back in to target cancerous cells.
This particular form of cell treatment sees the T cells being enhanced by the protein from the scorpion toxin.
In pre-clinical modelling the protein and the T cells have been shown to bind more broadly and specifically to glioblastoma cells than other prospective agents.
Modelling also suggests the newly enhanced cells demonstrate potent anti-tumour activity against glioblastoma whilst not showing any recognition of normal human cells and tissues, indicating a potentially optimal safety and efficacy profile.
City of Hope Professor and Chief, Division of Neurosurgery; Director, Brain Tumour Program, Department of Surgery, Dr Behnam Badie, said:
“This preliminary data is encouraging as it demonstrates safety with dual routes of administration. We now look forward to advancing the trial to higher dose levels which may provide more therapeutic benefit to patients.”
Chimeric has a number of cell therapies it is developing to broaden its suite of potential treatments.
The company is aiming to develop a further four new next-generation cell therapy assets with plans for phase one clinical trials to begin in 2023 in solid tumours and blood cancers.
The venture is one of the latest brought to market by bioentrepreneur Paul Hooper, chair of the $1.7 billion Imugene, Radiopharm Theranostics and Arovella Therapeutics. He also steered Viralytics into a $500 million takeover by medical behemoth Merck in 2018.
Also boosting the management team has been the recent appointment of respected haematologist Dr Steven Cha, as chief medical officer who will lead global clinical development efforts and medical affairs.
Chimeric appears to be making progress with multiple blue-chip partnerships with world leading medical institutions as it develops its arsenal of cancer fighting cell therapies. Success in any of the trials could lead to life-changing possibilities for the millions of cancer patients around the world.
Is your ASX-listed company doing something interesting? Contact: matt.birney@businessnews.com.au